Introduction: Patients with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) who fail multi-agent chemoimmunotherapy have a poor prognosis and a need for more treatment options. Loncastuximab tesirine (Lonca) comprises a humanized anti-CD19 antibody conjugated to a potent pyrrolobenzodiazepine dimer toxin. We present updated results from a phase 2 study of Lonca in patients with R/R DLBCL who had failed established therapies, including analysis of response in high-risk DLBCL subgroups.
Methods: Patients aged ≥18 years with R/R DLBCL who had failed ≥2 prior therapies were enrolled in this single-arm open-label phase 2 study (NCT03589469). Overall response rate (ORR) was assessed by independent reviewer according to the Lugano response criteria and duration of response (DoR) was defined as the time from earliest date of first response until the first date of either disease progression or death due to any cause. Pre-specified analyses of ORR and DoR by demographic and clinical characteristics were performed. Treatment-emergent adverse events (TEAEs) were reported for the all-treated population and by age group.
Results: In this study, 145 patients with DLBCL received Lonca (mean 4.3 cycles [range: 1-15]) and were evaluable for safety and efficacy. As of data cutoff (April 6, 2020), follow-up was ≥6 months since first dose.
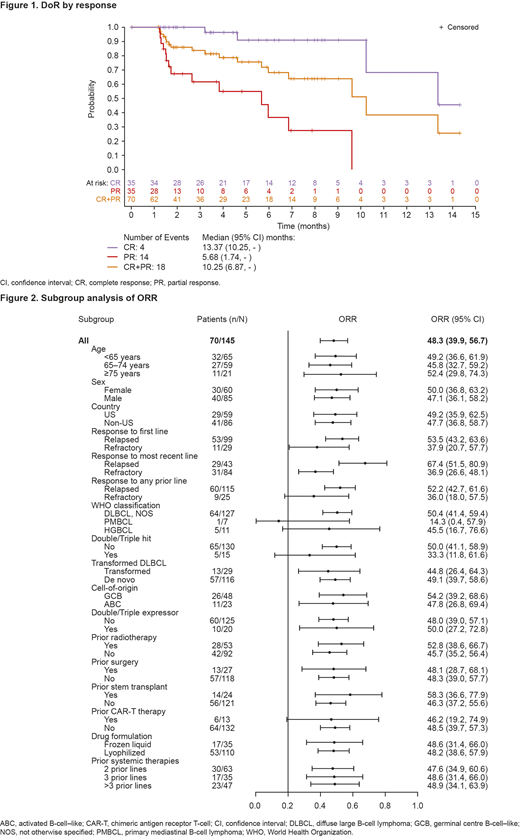
The ORR was 48.3%, and complete response rate (CRR) was 24.1%. Median DoR (mDoR) for the 70 responders was 10.3 months. For patients with a CR, mDoR was 13.4 months (Figure 1). Most patients responded after 2 treatment cycles; median time to first response was 41.0 days.
Responses, including CRs, were observed in several subgroups with higher-risk characteristics (Figure 2). ORR was 44.8% in patients with transformed disease, 45.8% in patients aged 65-74 years, and 52.4% in patients aged ≥75 years. For patients with double- or triple-hit DLBCL, ORR was 33.3% (notably all CRs). Patients who were refractory (no response) to first-line therapy, most recent therapy, or any prior therapy had ORRs of 37.9%, 36.9%, and 36.0%, respectively. Patients who had received a prior stem cell transplant (SCT) had a notable ORR of 58.3%. Lonca was also effective in patients who had received prior CD19-directed chimeric antigen receptor T-cell (CAR-T) therapy (ORR 46.2%).
Durable responses were noted in patients with high-risk characteristics. mDoR was not reached for patients with transformed disease. Older patients had a longer mDoR than younger patients (<65 years: 9.6 months; 65-74 years: 10.3 months; ≥75 years: 13.4 months). Patients with double- or triple-hit DLBCL had a mDoR of 13.4 months. mDoR was comparable for patients who were refractory to first-line therapy, most recent therapy, or any prior therapy (each 9.6 months) and for those who relapsed after first-line therapy, most recent therapy, or any prior therapy (each 10.3 months).
Following Lonca treatment, 15 patients received CD19-directed CAR-T therapy with an investigator-assessed ORR of 46.7% (6 CR; 1 partial response), and 9 patients proceeded to SCT as consolidation after responding to Lonca.
Overall, 143 (98.6%) patients had ≥1 TEAE and 105 (72.4%) patients had grade ≥3 TEAEs. The most common (≥25%) all-grade TEAEs were gamma-glutamyltransferase (GGT) increased (40.7%), neutropenia (39.3%), thrombocytopenia (33.1%), fatigue (27.6%), and anemia (26.2%). The most common (≥10%) grade ≥3 TEAEs were neutropenia (25.5%), thrombocytopenia (17.9%), GGT increased (16.6%), and anemia (10.3%). Treatment-related TEAEs leading to treatment discontinuation occurred in 24 patients (16.6%), most commonly GGT increased (14 patients; 9.7%). No increase in toxicity was seen in patients aged ≥65 years compared with younger patients.
Conclusions: Lonca had substantial single-agent antitumor activity in patients with R/R DLBCL. No new safety concerns were identified and no increase in toxicity was observed for patients aged ≥65 years. Encouraging and durable responses were observed in high-risk patient groups, including double- or triple-hit, transformed or refractory DLBCL, and notably in those who had progressed after prior CAR-T cell therapy. In addition, investigator-reported ORR indicates that many patients also respond to CAR-T therapy given after Lonca. Updated DoR data will be presented at the meeting.
Funding: This study is sponsored by ADC Therapeutics SA; clinicaltrials.gov/ct2/show/NCT03589469.
Caimi:Celgene: Speakers Bureau; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Genentech, ADC Therapeutics: Research Funding; ADCT, Kite Therapeutics, Genentech, Amgen, Verastem, TG Therapeutics, Bayer: Consultancy. Ai:ADC Therapeutics, Kymera: Membership on an entity's Board of Directors or advisory committees; Nurix Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding. Alderuccio:ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; OncLive: Honoraria; Puma Biotechnology: Other: Family member; Foundation Medicine: Other: Family member; Inovio Pharmaceuticals: Other: Family member; Oncinfo: Honoraria; Forma Therapeutics: Other: Family member; Agios Pharmaceuticals: Other: Family member. Ardeshna:Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi, Genzyme, AstraZeneca: Speakers Bureau; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; University College London (UCL)/UCL Hospitals (UCLH) Biomedical Research Unit: Other: Supported by this organisation; Beigene: Membership on an entity's Board of Directors or advisory committees. Hamadani:Takeda Pharmaceutical Company; Spectrum Pharmaceuticals; Astellas Pharma: Research Funding; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Janssen R&D; Incyte Corporation; ADC Therapeutics; Celgene Corporation; Pharmacyclics, Omeros, AbGenomics, Verastem, TeneoBio: Consultancy; Sanofi Genzyme, AstraZeneca: Speakers Bureau. Hess:ADC Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; BMS, AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Kahl:Roche Laboratories Inc: Consultancy; Genentech: Consultancy; ADC Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene Corporation: Consultancy; AstraZeneca Pharmaceuticals LP: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Acerta: Consultancy, Research Funding; BeiGene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics LLC: Consultancy. Radford:GlaxoSmithKline: Current equity holder in publicly-traded company, Other: Spouse; AstraZeneca: Current equity holder in publicly-traded company, Other: Spouse; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pfizer: Research Funding; ADCT: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria; Seattle Genetics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Stathis:Member of the steering committee of the trial of this abstract: Other; Loxo: Honoraria, Other, Research Funding; Cellestia: Research Funding; PharmaMar: Other: Travel Grant; Abbvie: Other: Travel Grant; ADC Therapeutcis: Other, Research Funding; MEI Pharma: Other, Research Funding; Novartis: Other, Research Funding; Bayer: Other, Research Funding; Merck: Other, Research Funding; Roche: Other, Research Funding; Pfizer: Other, Research Funding. Zinzani:Servier: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Portola: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; MSD: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Verastem: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celltrion: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen-Cilag: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sandoz: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; EUSA Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Therapeutics, Inc.: Honoraria, Speakers Bureau; Kirin Kyowa: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Kyowa Kirin: Consultancy, Speakers Bureau; Eusapharma: Consultancy, Speakers Bureau. Feingold:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company. Ungar:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company. Qin:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company. He:ADC Therapeutics: Current Employment, Current equity holder in publicly-traded company. Carlo-Stella:Servier, Novartis, Genenta Science srl, ADC Therapeutics, F. Hoffmann-La Roche, Karyopharm, Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics and Rhizen Pharmaceuticals: Research Funding; Bristol-Myers Squibb, Merck Sharp & Dohme, Janssen Oncology, AstraZeneca: Honoraria; Boehringer Ingelheim and Sanofi: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal